To know about what are valence electrons, we should first known about atom because it is all about the structure of atom. An atom is formed of electrons, protons and neutrons. An electron is a basic element of an atom. Nucleus of the atom contains neutrons, which are uncharged particles and protons which are positively charged. Electrons are present around the nucleus of the atom. These are negatively charged. The electrons situated in the inner shell of an atom are the core electrons of an atom. The number of electrons and protons are always the same. Electrons are arranged around the nucleus of an atom. This arrangement is sometimes called as electrons cloud.
Electrons revolve around the nucleus just like the sun rotates around the planets. Every kind of atom (microscopic) contains electrons. Hydrogen atom contains smallest number of electrons in it i.e. one proton and one electron. Uranium atom (largest atom) contains total 92 electrons.
Now what are valence electrons? So valence electrons are those special electrons which are placed in the most exterior valence shell of an atom. And now the question is why they are special? So the answer is because whenever there is an interaction between any two atoms, the electrons present in their outermost shell are the ones which first communicate with each other. When they interact they actually determine the reaction of an atom in a chemical reaction. They are responsible for incitement of a chemical reaction.
Valence electrons are very small subatomic parts which come transversely during the study of an atom of the element. An atom’s electrons can be earned or lost during a chemical reaction. With the help of Lewis structure we can track valence electrons and also presume the bonding. They are accountable for the intercommunication between atoms and also the construction of chemical bonds. An atom has many electrons but only valence electrons can take part in the process of chemical bond formation and molecule development.
Key characteristics of valence electrons:
- They exist in the outermost shell only for main group elements.
- They can also exist in inner shell for transition metals.
- They can absorb or release energy in the form of a photon.
- These electrons also determine an atom’s electric conductivity.
They are the electrons of the outermost shell of an atom which are not filled. Basically “valence” means number of electrons, the atom has to gain or lose to achieve nearby inert gas or noble gas electronic configuration. Elements having 8 valence electrons are called as noble gases or inert gases. These gases are not inclined to have chemical reactions with any other elements of periodic table group. The number of valence electrons of an element is similar to number of the group of that element in the periodic table. Hence to determine an element’s group in the periodic table you need to know the number of valence electrons in that element.
Let’s take some examples to visualize valence electrons:
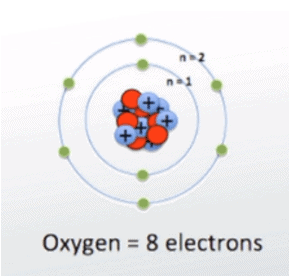
If we take oxygen, it has 6 electrons in its outer most valence shell. That means oxygen has 6 valence electrons.
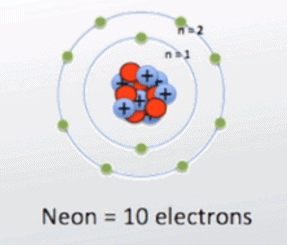
Now take neon, it has 8 electrons in its outermost valence shell so it has 8 valence electrons.
Every shell of an atom has certain amount of sub shells such as s, p, and d having certain orbital. Each orbital hold 2 electrons.
- First shell of an atom has 1 sub shell (s) having 1 orbital that can hold 2 electrons.
- Second shell has 2 sub shells (s and p). P has 3 orbital which can hold 6 electrons. So, second shell can hold a total 8 electrons.
- Third shell has 3 sub shells (s, p and d). D has 5 orbital which can hold 10 electrons. That means third shell can hold total 18 electrons.
Importance of valence electrons:
They are very important because they allow intense revelation about the chemical properties of an element including the nature (electronegative / electropositive), bond order and how many bonds can be created between two atoms. Covalent bonds are created due to sharing of electrons found in the exterior shell and the number of these electrons signifies that how many bonds can be formed. You can calculate the number of valence electrons in an element if you know the last number of electronic configuration of that element.
Function of valence electrons:
These are most exposed electrons of an atom and act as a protective barrier. They are in the highest energy level. These are the most involved electrons in chemical reaction and can be transferred very easily. The role of valence electrons is to transfer between the atoms to make the atom stable. The reactivity or inertness of an atom solely depends on its number of valence electrons. This is the mail thing that makes valence electrons an essential component of an atom. An atom cannot exist without it.
How to determine valence electrons:
Amount of valence electrons in an element can be calculated with the help of periodic table and electronic configuration of the element. Periodic table is much easier than finding the electric configuration. Elements are categorized in periodic table group. All elements are very neatly arranged in periodic table. You can see the elements from left to right are arranged in ascending order of their atomic numbers. Just find the position of element (whose valence electrons you want determine) in the periodic table. There are 4 main categories of elements including main group elements, transition elements, actinides and lanthanides.
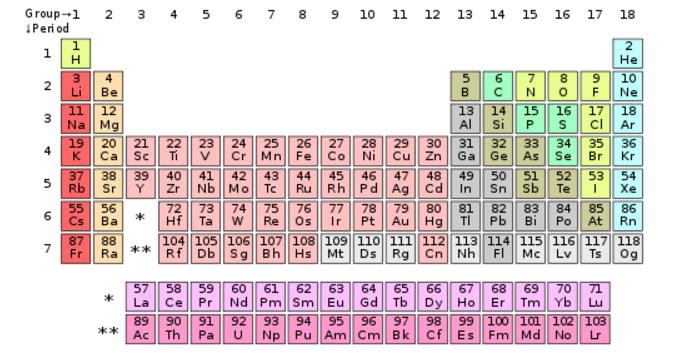
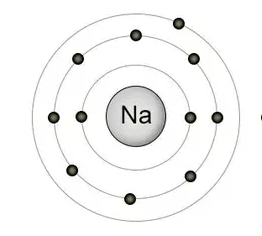
Let’s take element “sodium” for example. Sodium (Na) exists in group 3 and period 1 that means it has total three shells and its valence shell has single electron.